Modeling Aqueous Secondary Organic Aerosol Formation: GAMMA, simpleGAMMA, and Properties Modules
Overview
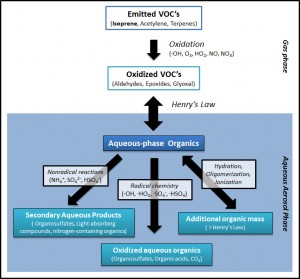
GAMMA (Gas-Aerosol Model for Mechanism Analysis) is a photochemical box model that features detailed, coupled gas- and aqueous aerosol-phase chemistry. GAMMA was originally designed to allow speciated prediction of secondary organic aerosol (SOA) and organosulfate formation in the aqueous aerosol phase under ambient or laboratory conditions (McNeill et al., 2012). It has also been applied to study inorganic aqueous chemistry in aerosols and aqueous chemistry, including SOA formation, in cloudwater.
simpleGAMMA is a reduced version of GAMMA suitable for coupling with larger scale atmospheric chemistry models (Woo and McNeill (2015)).
For questions about GAMMA or simpleGAMMA, or to request the code, contact us.
GAMMA: Mechanism
The original chemical mechanism and governing equations of GAMMA are described in McNeill et al (2012). A complete listing of the gas and aqueous phase chemical mechanisms and parameters used can be found in the supporting information here. GAMMA currently includes the gas-phase photochemistry of isoprene, acetylene, toluene, and xylenes, as well as the inter-phase transport and subsequent aqueous aerosol-phase processing of their water-soluble oxidation products. GAMMA follows over 160 gas- and aerosol-phase species and more than 260 reactions. Both GAMMA and simpleGAMMA are written for MATLAB and use the stiff solver ode15s.
Note that development of the GAMMA mechanism is an active area of research for our group and is continuously being updated and expanded. Recent updates and additions to GAMMA include:
- Expanded and updated gas phase chemistry (C1-C3)
- Detailed S(IV) to S(VI) (sulfate formation) chemistry
- Transition metal ion chemistry: Fenton, Clustering, sulfate formation
- Photosensitizer chemistry (e.g. Tsui et al. 2017, Tsui and McNeill 2018)
- Reactive uptake option
- Aqueous-phase reactions of IEPOX with NH4+, and updated IEPOX Henry’s Law constant (Budisulistiorini et al. 2015)
- Detailed H+ kinetics/Online thermodynamics (ISORROPIA/ISOCALL)
- Salting effects for glyoxal (Curry et al. 2018)
simpleGAMMA
Results from McNeill et al. (2012) indicated that aqueous aerosol SOA mass and organosulfate generation were dominated by isoprene-derived epoxydiol (IEPOX) uptake and processing under low-NOx conditions, or dark uptake of glyoxal under high-NOx conditions. This suggested a natural approach for reducing the GAMMA mechanism to a computationally light version, useful for coupling with large-scale atmospheric chemistry models. simpleGAMMA tracks only 4 aqueous species (IEPOX, IEPOXOS, 2-methyltetrol, and glyoxal) and only 2 species are passed between the gas and aqueous phases (glyoxal and IEPOX). In test runs, agreement to within 25% of the aqueous aerosol SOA mass predicted by the full model is acheived (20% for low-NOx). Details regarding simpleGAMMA can be found in Woo and McNeill (2015).
Physical Property Modules
Aqueous-phase SOA formation can potentially impact the optical properties and surface tension of the seed aerosol. We have developed modules based on parameterizations of our laboratory studies to predict the UV-visible light absorption and surface tension based on GAMMA output. Details can be found in Woo et al. (2013).
References
“Aqueous-phase Secondary Organic Aerosol and Organosulfate Formation in Atmospheric Aerosols: A Modeling Study” VF McNeill, JL Woo, DD Kim, AN Schwier, NJ Wannell, AJ Sumner, JM Barakat. Environ. Sci. Technol., 46 (15), 8075–8081 (2012). link to paper
“Aqueous aerosol SOA formation: Impact on aerosol physical properties” JL Woo, DD Kim, AN Schwier, R Li and VF McNeill. Faraday Discuss., 165, 357-367 (2013). link to paper
“Modeling the surface tension of complex, reactive organic-inorganic mixtures” AN Schwier, GA Viglione, Z Li, and VF McNeill. Atmos. Chem. Phys., 13, 10721-10732 (2013). link to paper
“Climate-Relevant Physical Properties of Molecular Constituents Relevant for Isoprene-Derived Secondary Organic Aerosol Material” MA Upshur, BF Strick, VF McNeill, RJ Thomson, FM Geiger. Atmos. Chem. Phys., 14, 10731-10740 (2014). link to paper
“Model Analysis of Secondary Organic Aerosol Formation by Glyoxal in Laboratory Studies: The Case for Photoenhanced Chemistry” AJ Sumner, JL Woo, VF McNeill. Environ. Sci. Technol., 48 (20), 11919-11925 (2014). link to paper
“simpleGAMMA 1.0 – A reduced model of secondary organic aerosol formation in the aqueous aerosol phase (aaSOA)” JL Woo and VF McNeill, Geosci. Model. Dev. 8, 1821-1829 (2015) link to paper
“Examining the effects of anthropogenic emissions on isoprene-derived secondary organic aerosol formation during the 2013 Southern Oxidant and Aerosol Study (SOAS) at the Look Rock, Tennessee, ground site” S.H. Budisulistiorini, X. Li, S.T. Bairai, J. Renfro, Y. Liu, Y.J. Liu, K.A. McKinney, S.T. Martin, V.F. McNeill, H.O.T. Pye, A. Nenes, M.E. Neff, E.A. Stone, S. Mueller, C. Knote, S.L. Shaw, Z. Zhang, A. Gold, and J.D. Surratt, Atmos. Chem. Phys., 15, 8871-8888 (2015) link to paper
“Aqueous-phase mechanism for secondary organic aerosol formation from isoprene: application to the southeast United States and co-benefit of SO2 emission controls” E.A. Marais, D. J. Jacob, J. L. Jimenez, P. Campuzano-Jost, D. A. Day, W. Hu, J. Krechmer, L. Zhu, P.S. Kim, C.C. Miller, J.A. Fisher, K. Travis, K. Yu, T.F. Hanisco, G.M.Wolfe, H. L. Arkinson, H. O. T. Pye, K. D. Froyd, J. Liao, and V.F. McNeill (2016) . Atmos. Chem. Phys., 16, 1603-1618 (2016) link to paper
“Observational constraints on glyoxal production from isoprene oxidation and its contribution to organic aerosol over the Southeast United States” J. Li, J. Mao, K.E. Min, R.A. Washenfelder, S.S. Brown, J. Kaiser, F.N. Keustch, R. Volkamer, G.M. Wolfe, T.F. Hanisco, I. B. Polllack, T.B. Ryerson, M. Graus, J.B. Gilman, B.M. Lerner, C. Warneke, J.A. deGouw, A.M. Middlebrook, J. Liao, A. Welti, B.H. Henderson, V. F McNeill, S.H. Hall, K. Ullmann, L.J. Donner, F. Paulot, and L.W. Horowitz. J. Geophys. Res.-Atmos., 121(16), 9849-9861 (2016). link to paper
“Simulating Aqueous-Phase Isoprene-Epoxydiol (IEPOX) Secondary Organic Aerosol Production During the 2013 Southern Oxidant and Aerosol Study (SOAS) ” S.H. Budisulistiorini, A. Nenes, A. M.G. Carlton, J.D. Surratt, V.F. McNeill, H.O.T. Pye. Environ. Sci. Technol., 51 (9) 5026-5034 (2017). link to paper
“Modeling Photosensitized Secondary Organic Aerosol Formation in Laboratory and Ambient Aerosols” W.G. Tsui, Y. Rao, H.-L. Dai, V.F. McNeill, Environ. Sci. Technol. 51 (13) 7496-7501 (2017). link to paper
Mao, J.*, Carlton, A., Cohen, R.C., Brune, W.H., Brown, S.S., Wolfe, G.M., Jimenez, J.L., Pye, H.O.T., Ng, N.L., Xu, L., McNeill, V.F., Tsigaridis, K., McDonald, B.C., Warneke, C., Guenther, A., Alvarado, M.J., de Gouw, J., Mickley, L.J., Liebensperger, E.M., Mathur, R., Nolte, C.G., Portmann, R.W., Unger, N., Tosca, M., Horowitz, L.W. “Southeast Atmosphere Studies: learning from model-observation syntheses.” Atmos. Chem. Phys. 18, 2615-2651 (2018). link to paper
Tsui, W.G., McNeill, V.F.* “Modeling Secondary Organic Aerosol Production from Photosensitized Humic-like Substances (HULIS),” Environ. Sci. Technol. Lett., 5 (5), 255–259 (2018). link to paper
Curry, L.A., Tsui, W.G., McNeill, V.F.* “Technical Note: Recommended reactive uptake coefficients for uptake of glyoxal and methylglyoxal by atmospheric aerosols and cloud droplets” Atmos. Chem. Phys. 18, 9823-9830 (2018) link to paper
Tsui, W.G., Woo, J.L., McNeill, V.F.* “Impact of Aerosol-Cloud Cycling on Aqueous Secondary Organic Aerosol Formation” Atmosphere 10(11), 666 (2019) link to paper
Wang, S., Zhou, S., Tao, Y., Tsui, W.G., Ye, J., Yu, J.Z., Murphy, J.G., McNeill, V.F., “Organic Peroxides and Sulfur Dioxide in Aerosol: Source of Particulate Sulfate” Environ. Sci. Technol. 53(18), 10695-10704 (2019) link to paper
Fankhauser, A.M., Antonio, D.D, Krell, A., Alston, S.J., Banta, S., McNeill, V.F.* “Constraining the impact of bacteria on the aqueous atmospheric chemistry of small organic compounds” ACS Earth Space Chem. 3(8), 1845-1491 (2019) link to paper
“Urban pollution greatly enhances formation of natural aerosols over the Amazon rainforest” Shrivastava M.*, Andreae M.O., Artaxo P., Barbosa H.M.J., Berg L.K., Brito J., Ching J., Easter R.C., Fan J., Fast J.D., Feng Z., Fuentes J.D., Glasius M., Goldstein A.H., Alves E.G., Gomes H., Gu D., Guenther A., Jathar S.H., Kim S., Liu Y., Lou S., Martin S.T., McNeill V.F., Medeiros A., de Sa S.S., Shilling J.E., Springston S.R., Souza R.A.F., Thornton J.A., Isaacman-VanWertz G., Yee L.D., Ynoue R., Zaveri R.A., Zelenyuk A., Zhao C. Nature Communications 10(1), 1046 (2019) link to paper
Riva, M., Sun, J., McNeill, V.F., Ragon, C., Perrier, S., Rudich, Y. Nizkorodov, S.A., Chen, J., Caupin, F., Hoffmann, T.*, George, C.*, High Pressure Inside Nanometer-Sized Particles Influences the Rate and Products of Chemical Reactions, Environ. Sci. Technol., 55 (12) 7786-7793 (2021) link to paper
Tilgner, A., Schaefer, T., Alexander, B., Barth, M., Collett, Jr., J.L., Fahey, K.M., Nenes, A., Pye, H.O.T., Herrmann, H.*, McNeill,V.F.* Acidity and the multiphase chemistry of atmospheric aqueous particles and clouds. Atmos. Chem. Phys. 21, 13483-13536 (2021) link to paper
Barth, M.C., Ervens, B., Herrmann, H., Tilgner, A., McNeill, V.F., Tsui, W.G., Deguillaume, L., Chaumerliac, N., Carlton, A., Lance, S.M. Box Model Intercomparison of Cloud Chemistry. J. Geophys. Res.-Atmos. 126 (21) e2021JD035486 (2021) link to paper
This page has been viewed (963)